LIMS, SAP, SDMS, LES software
The Enterprise Solution from Thermo Scientific™
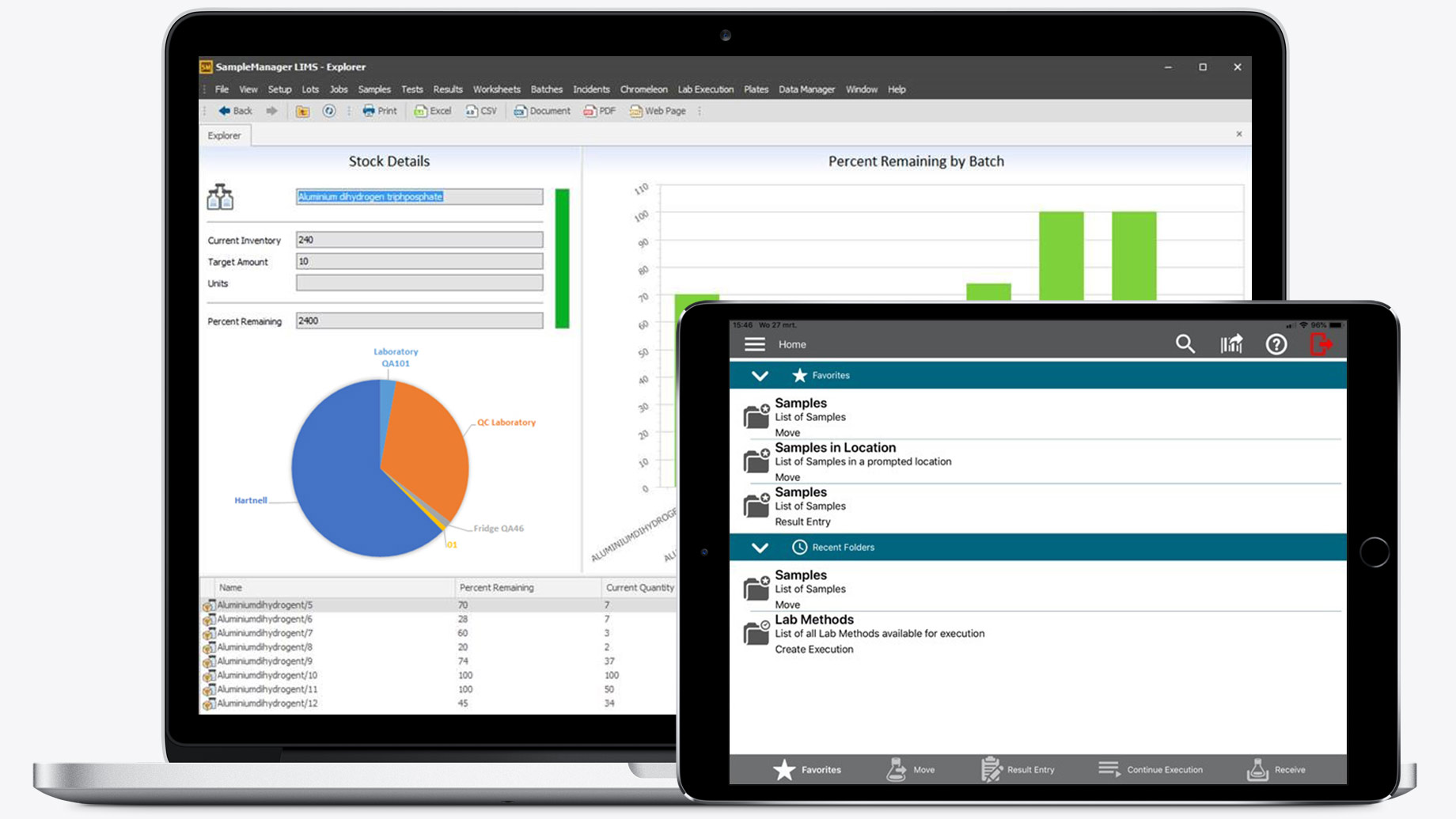
SampleManager LIMS™
Thermo Scientific SampleManager LIMS, SDMS, and LES software manages your laboratory, data, and process operations and connects to other enterprise systems, instruments, equipment, and your customers to ensure greater compliance and productivity throughout your organization.
Your Advantages at a Glance
Features SampleManager LIMS™
Compliance
Secure system access and controls to comply with the latest industry regulations such as GMP, ISO 17025 and FDA 21 CFR Part 11.
Data Analytics Solution
A set of out-of-the-box dashboards that reflect key business and lab insights, including resource availability, inventory information, site status, and lab performance, in a clear, interactive format.
Procedure Execution
Laboratory Execution System (LES) software guides analysts through each step of a procedure to ensure compliance with SOPs and captures all process history. Increase quality and compliance through repeatable execution of procedures and processes.
Functionality for Workflows
Users can quickly configure workflows according to their specific laboratory processes, automating decisions and actions. Workflows simplify initial system configuration and deployment, allowing laboratories to easily adopt new procedures.
Native Mobile Application
Allows access to LIMS and LES functions via a tablet, so you can “take the LIMS with you” and use it to run methods and acquire and process samples.
Centralized Data Management for Cromatography and MS
For even smoother operation and to ensure data integrity, SampleManager LIMS software is tightly integrated with Thermo Scientific Chromeleon CDS (chromatography data system) software.
Integration and Connectivity
LIMS software can connect to enterprise systems such as enterprise resource planning (ERP) systems and manufacturing execution systems (MES), as well as laboratory systems such as electronic laboratory notebooks (ELN) and chromatography data systems (CDS).
Cloud or On Premise Deployment
The option to deploy Thermo Fisher managed infrastructure including server, operating system and database, backup, disaster recovery and information security monitoring, reducing initial hardware and ongoing IT costs.
Electronic Laboratory Notebook ELN
A laboratory journal for analysts is integrated into LIMS, allowing users to add and link to a variety of data types and formats, such as rich text, spreadsheets and PDFs, and molecular structures.
Scientific Data Management System (SDMS)
Designed to comply with regulatory requirements, the software ensures that all raw data and associated metadata is secure and accessible for the entire data lifecycle, without the need for the original device software.
Contract Laboratory Testing and Contract Portals
Allows laboratories to manage all work elements. The job portal provides access for reviewing and approving quotes, registering samples, printing labels for shipping, and reviewing invoices and results.

Your contact person

Head of Sales – Customer Engagement Manager (CEM)
t + 49 6732 9490-57
e sales@uptodata.com
